 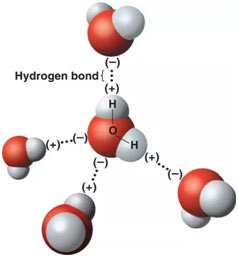
The symmetric − anion, reminiscent of the triiodide − anion, is found to be structurally equivalent to its cationic analogue + with N–I bond lengths of 2.26 Å. This list is followed by some char-acteristics that are observed in typical hydrogen-bonding environments.Ī series of LH halogen(I) complexes, where Z = saccharinato or phthalimido anions and LH = pyridinium, pyrazinium, tetrabutyl (TBA)- or tetramethylammonium (TMA) cations, were prepared, structurally characterized, and discussed as complexes consisting of a − anion with a three-center–four-electron (3c-4e) halogen bond, and a hydrogen-bonding (pyridinium or pyrazinium) or inert (TBA or TMA) cation. A list of criteria has been provided, and these can be used as evidence for the hydrogen bond formation. Considering the recent experimental and theoretical advances, we have proposed a new definition of the hydrogen bond, which emphasizes the need for evidence. Most important among them are the direct experimental evidence for a partial covalent nature and the observation of a blue-shift in stretching frequency following X–HY hydrogen bond formation (XH being the hydrogen bond donor and Y being the hydrogen bond acceptor). This debate has inten-sified following some important experimental results, especially in the last decade, which questioned the basis of the traditional view on hydrogen bonding. While its importance has been realized by physicists, chemists, biologists, and material sci-entists, there has been a continual debate about what this term means. 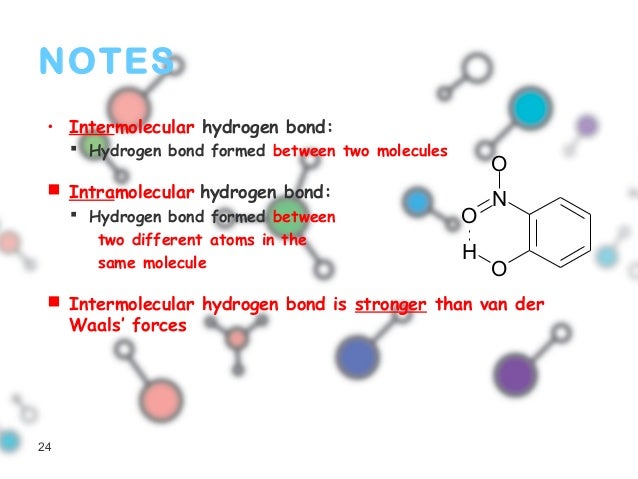
The term "hydrogen bond" has been used in the literature for nearly a century now. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
